Decoding the dark matter of our DNA: Study links genetic variants to blood pressure regulation
Scientists at The Hospital for Sick Children (SickKids) are delving deep into the non-coding genome to unravel the complex genetics that underlie blood pressure regulation and hypertension (high blood pressure)—the leading cause of cardiovascular disease affecting 1.25 billion people worldwide.
While the human genome comprises coding and non-coding DNA, the latter has long been overshadowed by its protein-coding counterpart and is commonly dismissed as “junk DNA.” In recent years scientists have identified several genomic regions that are associated with high blood pressure, including differences in DNA (also known as ‘variants’) in the non-coding genome.
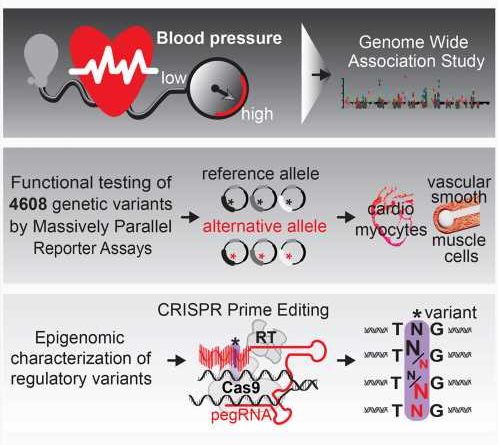
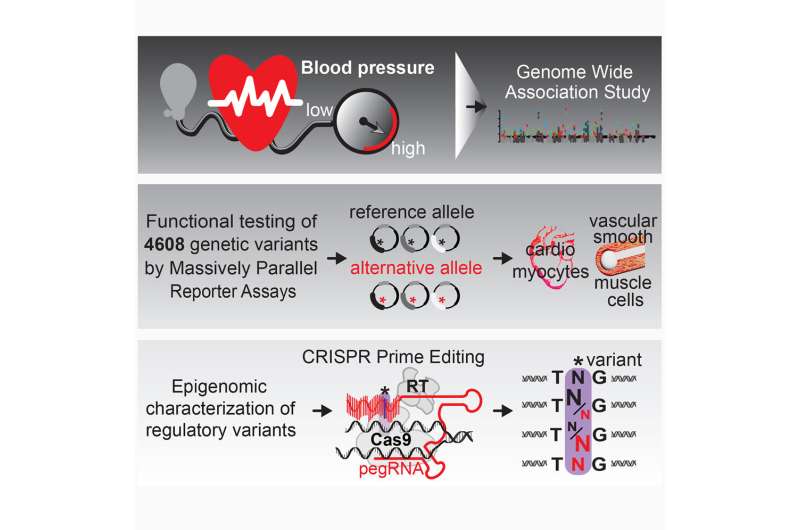
A research team led at SickKids by Dr. Philipp Maass, a Scientist in the Genetics & Genome Biology program, set out to understand how these variants likely regulate blood pressure genes and in doing so describe regulatory processes in the genome for blood pressure gene regulation. Their findings, recently published in Cell Genomics, offer a compelling glimpse into how these genetic sequences influence regulation of genes associated with high blood pressure.
“This research unveils, for the first time, the intricate connection between how variants in the non-coding genome affect genes that are associated with blood pressure and with hypertension,” explains Maass. “What we’ve created is a kind of functional map of the regulators of blood pressure genes that can not only inform future investigations into cardiovascular genomics, but that also presents a framework that can be applied to study other genetic conditions.”
Analysis of thousands of genetic variants finds connection to blood pressure regulation
Despite constituting 98 percent of our genetic material, the non-coding genome does not actively produce proteins. Instead, non-coding sequences regulate coding genes in various ways.
While more traditional genome-wide association studies (GWAS) are used to identify associations between genetic traits and specific diseases, they are unable to describe how genetic variants act and influence nearby genes. Findings from GWAS have informed much research on the function of variants in the coding genome, but very few have expanded to include variants on non-coding regions of the genome.
Maass and his team used massively parallel reporter assay (MPRA) technology at SickKids and computational expertise from Dr. Marta Melé at the Barcelona Supercomputing Center to examine genetic variants in the non-coding genome at a massive scale and identify how they likely regulate blood pressure genes. By leveraging stem cell expertise from Dr. James Ellis, a Senior Scientist in the Developmental & Stem Cell Biology program, the team was able to study the genetic variants in human relevant heart cells.
The study, which reviewed more than 4,600 genetic variants with the high throughput technology, represents one of the largest endeavors to date investigating and assigning function to the non-coding genome. Surprisingly, Maass notes, the analysis revealed high densities of variants located at genes tied to blood pressure regulation.
“With the growing adoption of whole genome sequencing, we can find thousands of new variants in a single genome,” says co-author Dr. Seema Mital, Staff Cardiologist, Senior Scientist in the Genetics & Genome Biology program, and Scientific Lead, Ted Rogers Centre for Heart Research and Head of Cardiovascular Research at SickKids. “Discerning which variants may be linked to disease holds the potential to enhance the utility of genome sequencing in clinical settings.”
Findings could inform precision medicine approaches for cardiovascular health
For the research team, uncovering the function of these genetic variants is an important step toward the future of Precision Child Health, a movement at SickKids to deliver individualized care for every patient. They hope information about these variants and their role in blood pressure regulation could one day be used to help clinicians predict which children may develop high blood pressure and provide appropriate interventions earlier.
“The variants we have characterized in the non-coding genome could be used as genomic markers for hypertension, laying the groundwork for future genetic research and potential therapeutic targets for cardiovascular disease,” notes Maass.
Beyond the impact on cardiac care, the findings may also inform similar approaches to other conditions with an underlying genetic component.
“Our research allowed us to peer into the ‘dark matter’ of our DNA, revealing insights that could be used as a roadmap to explore the genomic architecture behind various genetic traits. By combining different genomic, biochemical, and computational methodologies, this innovative approach holds promise for redefining our understanding of the regulatory role played by the non-coding genome in child health,” says Maass, who holds a Canada Research Chair Tier in Non-coding Disease Mechanisms.
More information:
Winona Oliveros et al, Systematic characterization of regulatory variants of blood pressure genes, Cell Genomics (2023). DOI: 10.1016/j.xgen.2023.100330
Journal information:
Cell Genomics
Source: Read Full Article