New PET radiotracer proven safe and effective in imaging malignant brain tumors
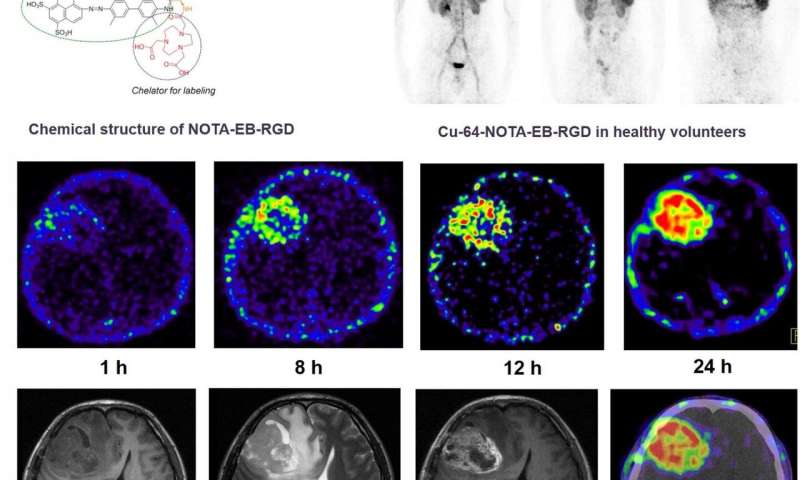
A first-in-human study presented at the Society of Nuclear Medicine and Molecular Imaging 2020 Annual Meeting has demonstrated the safety, favorable pharmacokinetic and dosimetry profile of 64Cu-EBRGD, a new, relatively long-lived PET tracer, in patients with glioblastomas. The radiotracer proved to be a superior, high-contrast imaging diagnostic in patients, visualizing tumors that express low or moderate levels of αvβ3 integrin with high sensitivity.
Glioblastoma is the most common and most aggressive primary malignant brain tumor in adults, with 17,000 diagnoses annually. It is a highly diffuse and invasive disease that is personally devastating and virtually incurable. Once diagnosed, most patients survive less than 15 months, and fewer than five percent survive five years.
The 64Cu-EBRGD radiotracer presented in this study has several unique qualities. The peptide sequence Arg-Gly-Asp (RGD) specifically targets the cell surface receptor αvβ3 integrin, which is overexpressed in glioblastomas. To slow clearance, Evans Blue (EB) dye, which reversibly binds to circulating albumin, is bound to RGD, significantly enhancing target accumulation and retention. The addition of the 64Cu label to EBRGD provides persistent, high-contrast diagnostic images in glioblastoma patients.
This first-in-human, first-in-class study included three healthy volunteers who underwent whole-body 64Cu-EBRGD PET/CT. Safety data—including vital signs, physical examination, electrocardiography, laboratory parameters and adverse events—were collected after one day and after one week. Regions of interest were drawn, time-activity curves were obtained and dosimetry was calculated. Two patients with recurrent glioblastoma also underwent 64Cu-EBRGD PET/CT. Seven sets of brain PET and PET/CT scans were obtained over two consecutive days. Tumor-to-background ratios were calculated for the target tumor lesion and normal brain tissue. One week after radiotracer administration, the patient underwent surgical treatment, and immunohistochemical staining of tumor samples was performed.
64Cu-EBRGD was well-tolerated in patients with no adverse symptoms immediately or up to one week after administration. The mean effective dose of 64Cu-EBRGD was very similar to the effective dose of an 18F-FDG scan. Injection of 64Cu-EBRGD to the patients with recurrent glioblastoma showed high accumulation at the tumor with continuously increased tumor-to-background contrast over time. Post-operative pathology revealed World Health Organization grade IV glioblastoma, and immunohistochemical staining showed moderate expression of the αvß3 integrin.
“In this study, we have demonstrated a potential radiotheranostic agent that is safe, sensitive and highly selective in humans, which infers a future diagnostic tool and breakthrough targeted radiotherapy for glioblastoma patients,” said Jingjing Zhang, MD, Ph.D., of Peking Union Medical College Hospital, Beijing, China. “We believe this innovative use of 64Cu-EBRGD will significantly improve therapeutic efficacy and patient outcomes.”
Source: Read Full Article
