flagyl mantar yaparm
A specific molecule excreted by methane-oxidizing bacteria could be most suitable for therapy against Wilson’s disease (WD). This is what researchers led by Helmholtz Munich scientist Hans Zischka have found in a new study. In a WD rat model, the molecule called ARBM101 lowered copper in the liver to normal physiological levels within a week—much more efficiently than current drugs in clinical practice. The research results have now been published in the journal Gastroenterology.
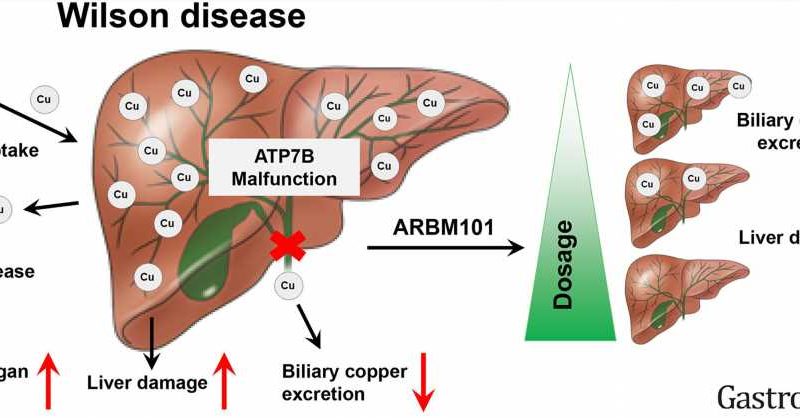
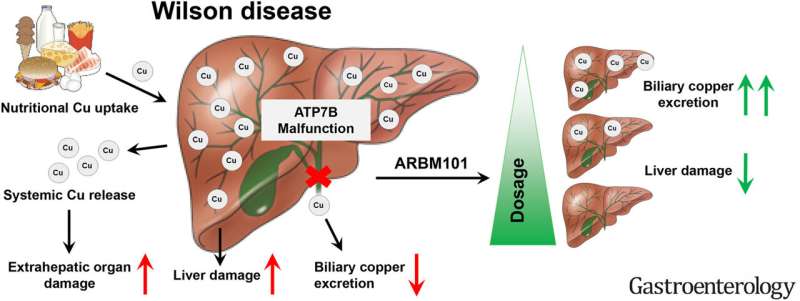
Wilson’s disease is a rare, hereditary disorder of copper metabolism in the liver. Due to defective biliary excretion, patients accumulate copper, especially in the liver and brain. If left untreated, the disease is deadly. However, can i take cataflam with ibuprofen with early diagnosis and consistent drug therapy, affected individuals may have a normal life expectancy.
A handful of therapeutic agents, mostly copper-binding chelators, are used in current treatments for the disease. They are designed to bind excess liver copper and promote its excretion, typically via the urine. While this can gradually reduce the amount of copper, levels do not approach normal physiological levels. Consequently, lifelong daily dosages are required to prevent disease progression. However, a lack of patient adherence or adverse drug reactions can lead to serious problems and eventual treatment failure.
Agent from bacteria as a new therapeutic option
Compared to synthetic chelators, methane-oxidizing bacteria are true copper hunters. Due to their vital need for copper, they have optimized their method of safe and effective copper mobilization: they produce and excrete the molecule “methanobactin” (MB), which binds copper very efficiently from the environment, to be taken up subsequently. The different bacterial strains each produce their “typical” methanobactin, so that we are dealing with an entire family of so-called “chalcophores” (ancient Greek χαλκός chalkós for copper). Despite structural differences, all representatives are highly copper-specific and mobilize the essential transition metal. Consequently, it may be suitable for copper mobilization/excretion in Wilson’s disease that would open new therapeutic options.
An international research team from Germany, Denmark, Portugal, the Republic of Korea, and the U.S. have now investigated groups of methanobactins regarding efficient copper excretion in a stringent rat model for Wilson’s disease. Led by Hans Zischka, head of the Mitochondrial Toxicology research group at the Institute of Molecular Toxicology and Pharmacology at Helmholtz Munich and at the Institute of Toxicology and Environmental Hygiene at the Technical University Munich (TUM), they placed a particular focus on one MB named ARBM101.
To test treatment safety and duration of efficacy, the research team developed a new treatment consisting of weekly applications followed by month-long treatment breaks. The results showed that the active ingredient ARMB101 lowered copper in the liver to normal physiological levels within eight days, eliminating the need for continuous treatment. In addition, the active ingredient was very well tolerated in the model and no negative effects in other organs were observed.
Short treatment, long breaks
The research results are of particular importance because they open up new treatment options for Wilson’s disease. The investigated compound ARBM101 not only eliminates excess liver copper safely and efficiently but also significantly shortens the therapy period and thus even allows for resting periods between treatments.
“A key finding of our work is that molecules for the treatment of excess metal diseases must precisely not ‘only’ be very specific for the metal in question, but must also keep them mobile upon binding, shield their reactivity, and bring them safely to excretion. Nature shows us this in a masterly way in the form of methanobactins,” says Hans Zischka, lead of the study.
More information:
Claudia Einer et al, ARBM101 (Methanobactin SB2) drains excess liver copper via biliary excretion in Wilson Disease rats, Gastroenterology (2023). DOI: 10.1053/j.gastro.2023.03.216
Journal information:
Gastroenterology
Source: Read Full Article